
Marine Science
Unit #2 explains more about water, or the properties of water. Below are some of my class notes I took on Wednesday, September 18. Also below are my notes on a lab I did the day after. The other notes are from the other labs I completed in my class.
- The Properties of Water Lab + Notes ✔
- Introduction to Density Lab ✔
- Rainbow Density Lab ✔
- Bill Nye - Buoyancy Notes ✔
- Phytoplankton Lab ✔
Unit #2 explains more about water, or the properties of water. Below are some of my class notes I took on Wednesday, September 18. Also below are my notes on a lab I did the day after. The other notes are from the other labs I completed in my class.
Properties of Water:
Water Notes
What is Water?
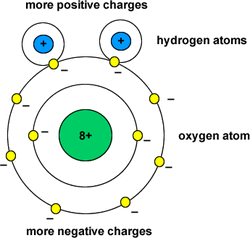
Two hydrogen atoms, one oxygen atom (H2O). An atom is the smallest unit of matter that retains its properties. Atoms consist of protons, electrons, and neutrons. Protons have a positive charge. Electrons have no charge, or neutral charge. And Neutrons have negative charge. Atoms are usually neutrally charged because the number of protons and electrons are even. Uneven charged atoms are called ions.
When two hydrogen atoms and one oxygen atom combine to form a water molecule, each hydrogen atom shares its single electron with the oxygen atom and the oxygen atom shares one of its electrons with the hydrogen atom.
Water is a Polar Molecule
Shared electrons spend more time around the oxygen atom than the hydrogen atom, and the hydrogen atom carries a slightly positive charge (polar molecule) and the oxygen carries a slightly negative charge.
Water Notes
What is Water?
Two hydrogen atoms, one oxygen atom (H2O). An atom is the smallest unit of matter that retains its properties. Atoms consist of protons, electrons, and neutrons. Protons have a positive charge. Electrons have no charge, or neutral charge. And Neutrons have negative charge. Atoms are usually neutrally charged because the number of protons and electrons are even. Uneven charged atoms are called ions.
When two hydrogen atoms and one oxygen atom combine to form a water molecule, each hydrogen atom shares its single electron with the oxygen atom and the oxygen atom shares one of its electrons with the hydrogen atom.
Water is a Polar Molecule
Shared electrons spend more time around the oxygen atom than the hydrogen atom, and the hydrogen atom carries a slightly positive charge (polar molecule) and the oxygen carries a slightly negative charge.
Hydrogen Bonding - The attraction between weak positive hydrogen atoms and the weak negative oxygen atoms of a different molecule.
Hydrogen bonding is responsible for the unique property of water. (Hail Hydrogen!! Hail Hydrogen!!)
Hydrogen bonding is responsible for the unique property of water. (Hail Hydrogen!! Hail Hydrogen!!)
Water is a Universal Solvent
The polarity of water gives it the ability to dissolve or dissociate most compounds. Positive hydrogen side of H2O is attracted to the negative parts of the compound. Negative oxygen atoms are attracted to the positive atoms. This allows water to dissociate and break apart ionically charged substances.
Water is attracted to other water molecules (aka cohesion). The attraction of water molecules to other water molecules help create a "film" on the surface of bodies of water (aka surface tension). Water can also be attracted to other materials (aka adhesion).
States of Mater
Solid (ice), Liquid (water), and Gas (water vapor). Earth is the only planet in the solar system where water is found in liquid form. Temperature of a substance is a measure of average kinetic energy of the atoms and molecules measured in degrees. Heat is a measure of the total kinetic energy of the atoms and molecules measured in Calories. One calorie is equal to the amount of heat needed to raise the temperature of one gram of water by one degree Celsius. When you eat one hundred calories, you body needs energy to burn them off. When your body can't burn calories off, they store them off as fat.
The polarity of water gives it the ability to dissolve or dissociate most compounds. Positive hydrogen side of H2O is attracted to the negative parts of the compound. Negative oxygen atoms are attracted to the positive atoms. This allows water to dissociate and break apart ionically charged substances.
Water is attracted to other water molecules (aka cohesion). The attraction of water molecules to other water molecules help create a "film" on the surface of bodies of water (aka surface tension). Water can also be attracted to other materials (aka adhesion).
States of Mater
Solid (ice), Liquid (water), and Gas (water vapor). Earth is the only planet in the solar system where water is found in liquid form. Temperature of a substance is a measure of average kinetic energy of the atoms and molecules measured in degrees. Heat is a measure of the total kinetic energy of the atoms and molecules measured in Calories. One calorie is equal to the amount of heat needed to raise the temperature of one gram of water by one degree Celsius. When you eat one hundred calories, you body needs energy to burn them off. When your body can't burn calories off, they store them off as fat.

The pool is big, so it has more heat.

The boiling pot may be smaller in size but it has a higher temperature.
Effect of Heat on Water
High heat of vaporization - Hydrogen bonds must be broken so water can absorb a lot of heat without charging phase. It evaporates slowly and that causes the cooling effect of sweating. (Tada!!)
Water has to be boiled at a high temperature to be "hot". Most of the water on Earth is liquid, it's good for oceans. High Latent Heat of Fusion - a lot of heat is released when water freezes and a lot of heat is absorbed when ice melts. Water temperature is more stable than average temperature and water moderates climates. > When you sweat, it is good for you. The sweat glands make water ooze out of your skin, the water on your skin absorbs the heat from the body, making you cool down. (Don't wipe away your sweat when you exercise!!)
Water has a high specific heat, this means that water can absorb a large amount of heat before it begins to get hot. The high specific heat of water helps regulate the rate at which the air changes temperature, which is why the temperature change of seasons is so gradual, not sudden, especially near large bodies of water (like the oceans).
Density is a physical property of a substance that describes the degrees of compactness of a substance - how closely packed together the atoms are. > More packed together, more dense. Density - mass/volume - how much stuff in how much space? Usually substances become more dense (more compact) when they solidify. Water becomes less dense in its solid form (only substance to do this).
High heat of vaporization - Hydrogen bonds must be broken so water can absorb a lot of heat without charging phase. It evaporates slowly and that causes the cooling effect of sweating. (Tada!!)
Water has to be boiled at a high temperature to be "hot". Most of the water on Earth is liquid, it's good for oceans. High Latent Heat of Fusion - a lot of heat is released when water freezes and a lot of heat is absorbed when ice melts. Water temperature is more stable than average temperature and water moderates climates. > When you sweat, it is good for you. The sweat glands make water ooze out of your skin, the water on your skin absorbs the heat from the body, making you cool down. (Don't wipe away your sweat when you exercise!!)
Water has a high specific heat, this means that water can absorb a large amount of heat before it begins to get hot. The high specific heat of water helps regulate the rate at which the air changes temperature, which is why the temperature change of seasons is so gradual, not sudden, especially near large bodies of water (like the oceans).
Density is a physical property of a substance that describes the degrees of compactness of a substance - how closely packed together the atoms are. > More packed together, more dense. Density - mass/volume - how much stuff in how much space? Usually substances become more dense (more compact) when they solidify. Water becomes less dense in its solid form (only substance to do this).
