On Monday (the last day of September), my partner and I did a new lab, the Rainbow Salt Density Lab. The whole idea of this new lab was to make various saline solutions and layer them according to their respective densities. Down below is our work. :(|)
Rainbow Salt: An Investigation in Density
Materials:
• Scale/Balance
• One 50 mL Graduated Cylinder
• Two 150 mL Beakers
• Pipette
• A sheet of white background paper - in this case, we used our lab worksheets - This is used by putting it behind your completed work to see more clearly of the blended layers of the colors
• kosher Salt
• Food Coloring - Red, Orange, Yellow, Green and Blue
• Water
• Scale/Balance
• One 50 mL Graduated Cylinder
• Two 150 mL Beakers
• Pipette
• A sheet of white background paper - in this case, we used our lab worksheets - This is used by putting it behind your completed work to see more clearly of the blended layers of the colors
• kosher Salt
• Food Coloring - Red, Orange, Yellow, Green and Blue
• Water
Instructions:
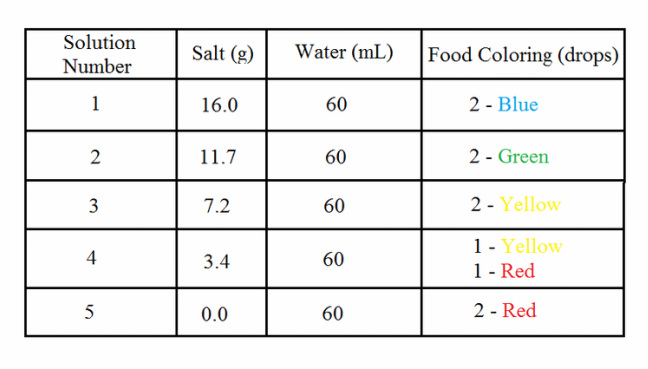
Prepare each solution by adding salt, water, and food coloring in the following amounts:
Prepare each solution by adding salt, water, and food coloring in the following amounts:

By carefully following the instructions from our lab worksheet, my partner and I got one graduated cylinder and two beakers. Next, we went to the sink to fill up our beakers with water. After finishing that up, we went to the salt station and measured 16.0 grams of salt for our first rainbow color. After that, we poured the 16.0 grams of salt into our beaker full of water. My partner then mixed the solution up.
As I watched, she mixed the salt into it dissolved... but there was still some salt left so I took the beaker and mixed it up 'til every grain of salt was dissolved completely. :P
"Really, Lieu?"
"Yes. The instructions clearly said to stir until ALL the salt dissolved."
As I watched, she mixed the salt into it dissolved... but there was still some salt left so I took the beaker and mixed it up 'til every grain of salt was dissolved completely. :P
"Really, Lieu?"
"Yes. The instructions clearly said to stir until ALL the salt dissolved."

After stirring the beaker, we got a pipette and got some blue food coloring, and slowly, one by one added three drops of the blue coloring into our beaker. Mrs. Ogo had told us earlier to add a few more drops of food coloring if we needed to, since the colors turned out too light. We did that and that's why we added one more drop of blue in.
After adding the drops, my partner began to stir once again, using a stirring rod. After stirring, we got such a beautiful blue color. :3
Down below is another picture of our beautiful blue. o u o
After adding the drops, my partner began to stir once again, using a stirring rod. After stirring, we got such a beautiful blue color. :3
Down below is another picture of our beautiful blue. o u o

After the process of stirring up the blue solution, we repeated the above steps with our other colors. We left the blue beaker next to our work station and I got up to get the water for the second beaker. Once I got the appropriate amount for the beaker, I gave my partner that beaker, and carefully paced over to the salt station and measured out 11.7 grams of salt. Once the measuring was done, I carefully carried the salt over to where my partner was sitting and poured in the salt. While she was stirring that beaker, I got my pipette and cleaned it up and got some green food coloring. I helped my partner stir the beaker solution up and squeezed five drops of green into that beaker. My partner once again began stirring.

Once the solution was perfected, we began adding the blue solution into our 50 mL graduated cylinder. I held the cylinder tightly with both my hands. I had tilted it at a 45 degree angle, so that when my partner squeezed the solutions in, the colors would be able to perfectly blend into each other. I also wanted the cylinder to be tilted because I had heard that it was much easier to do so like that. I also speak from experience. >:D She began with the beautiful blue. She squeezed some into a new pipette and began to pour in the blue, until it reached the 20 mL level. We didn't need to be careful on this one because it was the first color. You don't need to be careful and slow, because you aren't blending in colors... yet.
Next, my partner added the green into the cylinder, slowly but quickly, slowly but quickly. After doing that for quite some time, I told her to stop. I wanted to check if we had reached 40 mL yet. I checked and, we did reach it!
Next, my partner added the green into the cylinder, slowly but quickly, slowly but quickly. After doing that for quite some time, I told her to stop. I wanted to check if we had reached 40 mL yet. I checked and, we did reach it!

Next, we did the yellow. (We actually messed up doing yellow, we had gotten orange instead, so I got another beaker to do the new yellow). Once again, I gave a beaker to my partner and I (this time) ran over to the salt stations and measured out 7.2 grams of salt. I quickly, but gently, poured the salt into that beaker, and let my partner stir the solution up. I went and got the yellow food coloring with my pipette and dropped two into the beaker. After I did that, my partner quickly began stirring. (She finally got the idea to stir until everything was mixed and dissolved. Haha.)

After doing all that!, we repeated what we did with the beautiful blue and the vibrant green. I tilted the cylinder while my partner squeezed the yellow coloring in. If you look closely, you can see that the yellow is beginning to blend in with the vibrant green. Can you guys see it?
"Wait, if you two are doing that, who took the picture?"
Oh yeah. I'd also like to thank Marybelle Panilio for taking this picture. As y'all can see, my partner and I ran out of hands.... (Btw, click on her name to go to her website!!)
"Wait, if you two are doing that, who took the picture?"
Oh yeah. I'd also like to thank Marybelle Panilio for taking this picture. As y'all can see, my partner and I ran out of hands.... (Btw, click on her name to go to her website!!)

After pouring the urine-like yellow, the vibrant green and the beautiful blue, we made several changes to the orange-meant-to-be-yellow. We actually had to dump it. (WHAT A WASTE OF WATER!!!) When we made the salt measurement, we did 7.2 grams instead of 3.4 grams of salt. That's why we had to dump it. When I dumped all the orange out, I rinsed it out with clean water and began pouring different water into the beaker, to the right level and handed it to my partner. When she got it, I ran over to the salt station and measured out 3.4 grams of salt. I ran back over to my partner and poured in the salt. She began mixing. I ran over to get several drops of red food coloring and yellow food coloring. I returned to her side and waited. When she was done stirring, I dropped in one drop of red, and one drop of yellow. She began mixing that up. When it was done, we got yummy orange. (What? It was the only word I knew to describe orange at the time, stop judging me! xP)

Once again, I repeat the process. Run to get water, bring back to head quarters. Run to get salt measured to the right amount, 0.0 grams, run back to head quarters, run to get red food coloring, quickly walk back to head quarters. Input two drops of red, return pipette back to pipette bag, clean and looking semi-newish. Begin wait for mixed solution. Now is when things got different. I sat down next to my partner and held onto the cylinder with a tight grip with my hands and slowly tilted it to a 45 degree angle once again. The reason why I did the tilting slowly was because of the other blended color already inside of the cylinder. I didn't want our perfect layers to get mixed up. Next, my partner began squeezing in the orange... And then, the red. We had to hurry up because we'd had only ten minutes left to finish up. When suddenly, I had an idea.
"Hey, Sabrina. Let's wing it."
"What?"
"Let's hurry up and finish blending the orange and begin by red."
"Okay."
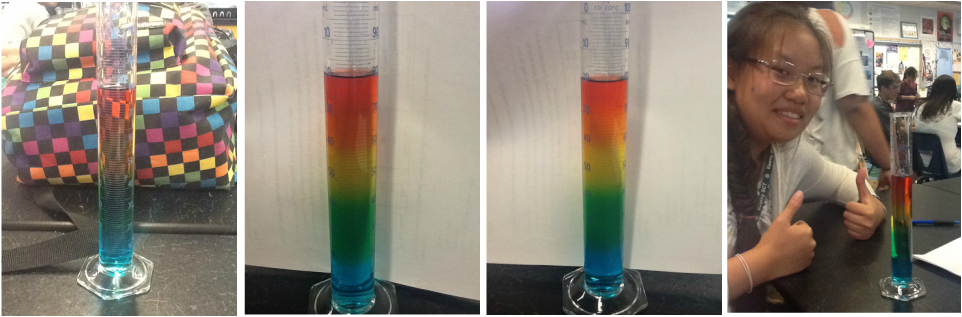
That's what we did. We found out later that we were supposed to get it to the 100 mL level but when I asked Mrs. Ogo about that, she said it was fine. Yay!! Next to this text, you can see the end result. You can see that ours is a bit short, but we still did it. We managed to finish it. Man, we were so happy when we finished it. Down below, you can see happy pictures of our rainbow lab. (≧▽≦)
"Hey, Sabrina. Let's wing it."
"What?"
"Let's hurry up and finish blending the orange and begin by red."
"Okay."
That's what we did. We found out later that we were supposed to get it to the 100 mL level but when I asked Mrs. Ogo about that, she said it was fine. Yay!! Next to this text, you can see the end result. You can see that ours is a bit short, but we still did it. We managed to finish it. Man, we were so happy when we finished it. Down below, you can see happy pictures of our rainbow lab. (≧▽≦)
Picture One: I took a pic of our cylinder in front of Dasia's backpack. I hope she doesn't mind. It's a pretty cool background, don't you think?
Picture Two and Three: Pix of our lab worksheet just behind the cylinder to show how mixed the solutions are. A friend had asked...
"How did you guys do that? It's so majestic! So well blended.." (That made my day... Hey. I think THE PERSON who said this might know... You know who you are PERSON.)
My response? I just nodded. Straight face. And just nodded.
My partner's response? "We did it through our blood, sweat and tears!! *ha* *haha* *haha*
What she said was true in a way. You know the feeling you get in your abdomen when you do push ups right? Yeah, I felt a small feeling just like that when I was hold the cylinder for my partner. I was also talking to her, laughing a the same time, and I didn't want to shake the cylinder, so my abdomen just squeezed and my heart had to pump more blood just so I would be concentrated... Yes, the real process of it is very hard, actually. IT'S NOT THAT SIMPLE, PEOPLE.
Picture Four: My fugly face next to our beautiful cylinder full of mixed solutions... Hurhur. C8 We each took turns taking pictures of each other with the cylinder. If you went onto my partner's weebly-web, maybe she'll have it up there too.
Picture Two and Three: Pix of our lab worksheet just behind the cylinder to show how mixed the solutions are. A friend had asked...
"How did you guys do that? It's so majestic! So well blended.." (That made my day... Hey. I think THE PERSON who said this might know... You know who you are PERSON.)
My response? I just nodded. Straight face. And just nodded.
My partner's response? "We did it through our blood, sweat and tears!! *ha* *haha* *haha*
What she said was true in a way. You know the feeling you get in your abdomen when you do push ups right? Yeah, I felt a small feeling just like that when I was hold the cylinder for my partner. I was also talking to her, laughing a the same time, and I didn't want to shake the cylinder, so my abdomen just squeezed and my heart had to pump more blood just so I would be concentrated... Yes, the real process of it is very hard, actually. IT'S NOT THAT SIMPLE, PEOPLE.
Picture Four: My fugly face next to our beautiful cylinder full of mixed solutions... Hurhur. C8 We each took turns taking pictures of each other with the cylinder. If you went onto my partner's weebly-web, maybe she'll have it up there too.
Calculations
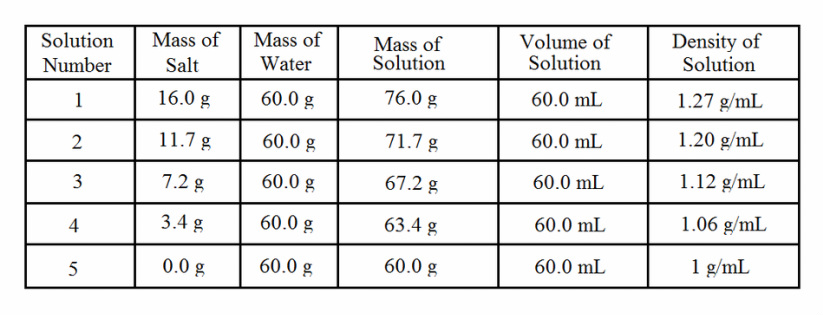
After doing the lab (and cleaning up), we were to complete our lab worksheet and write down what we saw and our analysis on salt density. We did our calculations and carried it to the hundredths place, if we needed to. I made a graph showing what we did.
After doing the lab (and cleaning up), we were to complete our lab worksheet and write down what we saw and our analysis on salt density. We did our calculations and carried it to the hundredths place, if we needed to. I made a graph showing what we did.
Analysis
This second graph is about what we had analyzed earlier in the lab. It relates to the graph above.
This second graph is about what we had analyzed earlier in the lab. It relates to the graph above.
Lab Report
Conclusion Questions:
If all of the solutions were made just with salt and water, why did they form different layers?
If you poured water into a graduate cylinder, it wouldn't have layers. They'd just blend together. But if you did the process of adding salt into different beakers, and poured/squeezed them into a different graduated cylinder, you'd get layers of density. But why do they have different layers? If you look back, you can see that the blue was added first. The green was added afterward. Why didn't the green just mix with the blue? Well, one, we poured the green in slowly at first, but that's not the real reason. It's because of the amount of salt you put in there. The "salinities". In the blue beaker, we added 16.0 grams of salt. That's the highest quantity of salt we put in any beaker. The water and salt solution in this beaker would sink to the bottom because it has the most density. The densest water will, of course, sink to the bottom. The green stayed just above the blue, because that has the second densest water level. Then yellow came next. That is the color that stayed in the middle. It wasn't really dense and it wasn't really not dense... It's in the between.. :o Then orange, then red. Red stayed at the top because it is not dense. At all. We did not pour in any salt. All we added was the red food coloring. This explanation is why they formed different layers.
The ocean contains water with different salinities. Where do you think the saltiest layers would be found? Why?
This is just like what I had explained before. The cylinder had different layers of water. Why? Each layer had an amount of salt in it. The layer with the most amount of salt sank. It stayed at the bottom. The layer with the least amount of salt stayed at the top. Does that same exact thing happen in the oceans too? Of course! If we take my analysis from up above, we can use it to explain where the saltiest layers can be found in the oceans. Since the blue had the most salt, it stayed at the bottom of the cylinder, correct? Now think about the ocean. If you think about a big amount of water in the ocean that has a lot of salinity, where do you think it would stay at? At the bottom! Water levels with the most salt will be the most dense. Water levels with the least amount of salt stays at the top. Everything depends on the quantity of salt. Take a look at the picture down below. (Idk if the stratification part has anything to do with density, but okay...)
Conclusion Questions:
If all of the solutions were made just with salt and water, why did they form different layers?
If you poured water into a graduate cylinder, it wouldn't have layers. They'd just blend together. But if you did the process of adding salt into different beakers, and poured/squeezed them into a different graduated cylinder, you'd get layers of density. But why do they have different layers? If you look back, you can see that the blue was added first. The green was added afterward. Why didn't the green just mix with the blue? Well, one, we poured the green in slowly at first, but that's not the real reason. It's because of the amount of salt you put in there. The "salinities". In the blue beaker, we added 16.0 grams of salt. That's the highest quantity of salt we put in any beaker. The water and salt solution in this beaker would sink to the bottom because it has the most density. The densest water will, of course, sink to the bottom. The green stayed just above the blue, because that has the second densest water level. Then yellow came next. That is the color that stayed in the middle. It wasn't really dense and it wasn't really not dense... It's in the between.. :o Then orange, then red. Red stayed at the top because it is not dense. At all. We did not pour in any salt. All we added was the red food coloring. This explanation is why they formed different layers.
The ocean contains water with different salinities. Where do you think the saltiest layers would be found? Why?
This is just like what I had explained before. The cylinder had different layers of water. Why? Each layer had an amount of salt in it. The layer with the most amount of salt sank. It stayed at the bottom. The layer with the least amount of salt stayed at the top. Does that same exact thing happen in the oceans too? Of course! If we take my analysis from up above, we can use it to explain where the saltiest layers can be found in the oceans. Since the blue had the most salt, it stayed at the bottom of the cylinder, correct? Now think about the ocean. If you think about a big amount of water in the ocean that has a lot of salinity, where do you think it would stay at? At the bottom! Water levels with the most salt will be the most dense. Water levels with the least amount of salt stays at the top. Everything depends on the quantity of salt. Take a look at the picture down below. (Idk if the stratification part has anything to do with density, but okay...)